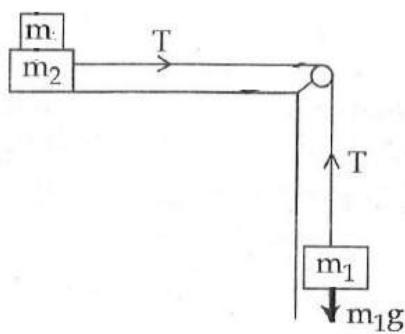
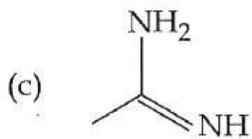
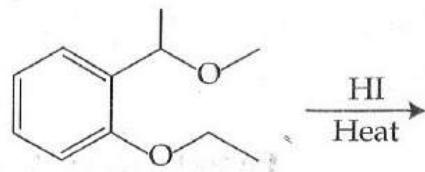
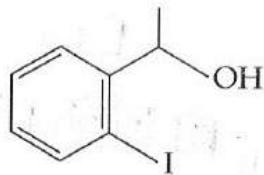
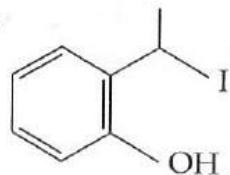
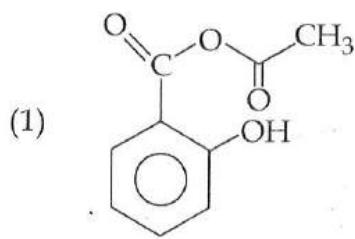
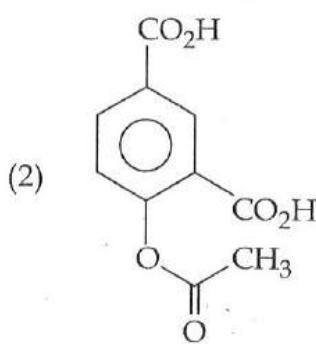
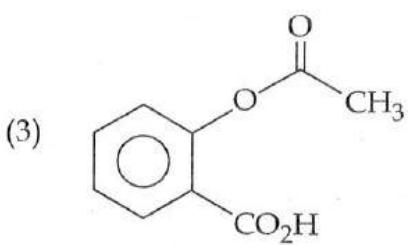
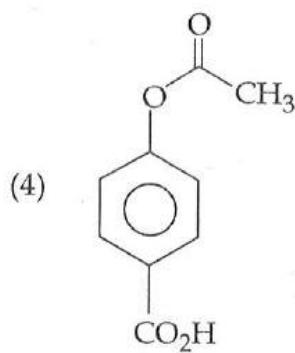
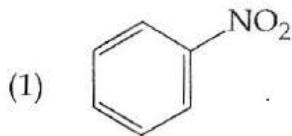
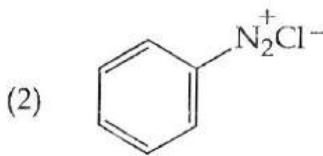
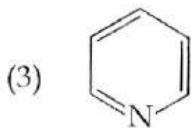
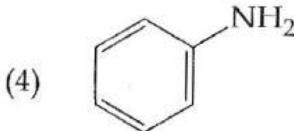
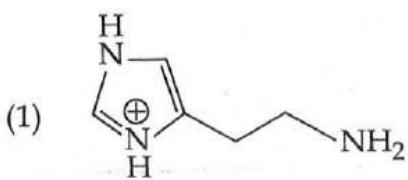
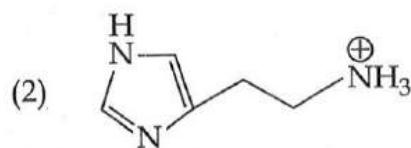
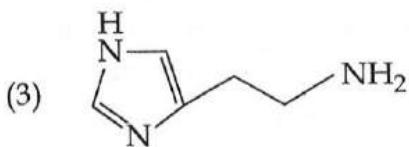
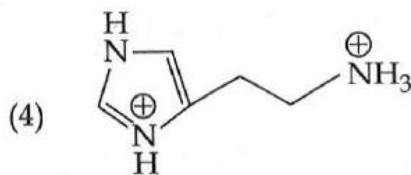
It is found that if a neutron suffers an elastic collinear collision with deuterium at rest, fractional loss of its energy is ; while for its similar collision with carbon nucleus at rest, fractional loss of energy is . The values of and are respectively:
- A.(0, 0)
- B.(0, 1)
- C.(.89, .28)Correct
- D.(.28, .89)